Chronic obstructive pulmonary disease (COPD) is a progressive disease, characterised by breathlessness and cough. Patients will typically experience exacerbations (some patients more so than others) and this negatively impacts disease progression, rates of hospitalisation and readmission, and health status. According to the Global Initiative for Chronic Obstructive Lung Disease 2019 report, the number of exacerbations in the previous year is the strongest predictor of a patient’s future exacerbation frequency, and the National Institute for Health and Care Excellence (NICE) reports that the rate of lung function decline is faster in the earlier stages of the disease, which can be modified by treatment.
In December 2018 NICE published an updated guideline for the diagnosis and management of COPD in over 16-year olds. This was followed by a further update in July 2019 which focused on inhaled triple therapy for stable COPD and systemic corticosteroids for managing exacerbations, where ‘triple therapy’ refers to the use of long acting beta agonist (LABA), long acting antimuscarinic agent (LAMA) and inhaled corticosteroid (ICS) together.
These updates supplement the existing recommendations on:
- diagnosing COPD using symptoms, spirometry and other tests
- managing stable COPD using nebulisers, oral therapy and pulmonary rehabilitation
- multidisciplinary management of stable COPD, including physiotherapy, occupational therapy, nutrition and palliative care
- managing exacerbations of COPD in primary care and in hospital.
Updates in the 2018 guideline were made on the following:
- Investigations, including incidental findings on CT scans: primary care review, advice for patients to return if respiratory symptoms appear, offering smoking cessation and discussion of potential lung cancer risk
- Prognosis:to avoid the use of a multidimensional index such as BODE to assess prognosis
- Inhaled therapies: to discuss risk of pneumonia and ICS use with patients, minimising the number and types of inhalers patients use, and ensuring they are trained on their use
- Oral phosphodiesterase 4 inhibitorsare mentioned in line with the associated 2017 technology appraisal
- Prophylactic antibiotics:recommendations made on (unlicensed) use of Azithromycin
- Oxygen therapy: eligibility of patients for, and risks of, prescription (including short burst and ambulatory use) and consideration for use in pulmonary hypertension. Noting this is not a treatment for breathlessness and not effective for isolated nocturnal hypoxaemia caused by COPD.
- Managing pulmonary hypertension and cor pulmonale:advice on optimised therapies
- Lung volume reduction (LVR) surgery and procedures:updated advice to increase uptake and access
- Self-management and exacerbation plans developed in collaboration with patients and carers.
People who are using long-acting bronchodilators outside of recommendations and whose symptoms are under control can continue with their current treatment until both they and their NHS healthcare professional agree it is appropriate to change.
The July 2019 update focused on promoting responsible prescribing of triple inhaled therapies to ensure patients gain maximal benefit from them, whilst limiting adverse effects and in the most cost-effective way. The guidance does not comment on the appropriateness or perceived benefit of using all three medicines in one single device or as separate inhalers, as this was outside of the scope and data for this was not reviewed.
Before starting LAMA+LABA+ICS, clinicians are advised to conduct a clinical review to ensure that:
- the person’s non-pharmacological COPD management is optimised and they have used or been offered treatment for tobacco dependence if they smoke
- acute episodes of worsening symptoms are caused by COPD exacerbations and not by another physical or mental health condition
- the person’s day-to-day symptoms that are adversely impacting their quality of life are caused by COPD and not by another physical or mental health condition.
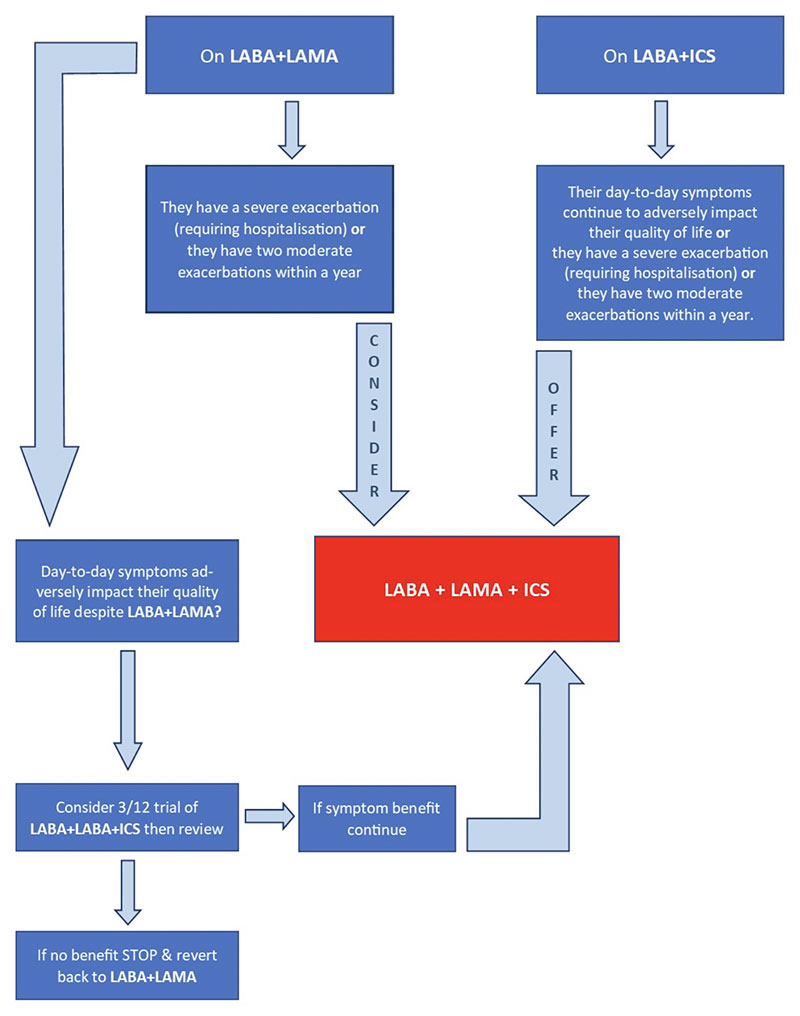
NICE also recommend documenting the reason for continuing ICS use in clinical records and review this at least annually. The recommendation on systemic use of corticosteroids was also reviewed and updated: 30mg oral prednisolone is to be offered for five days duration for exacerbations. This is aligned with five-day prescribing of antibiotics for COPD exacerbations.
The full guideline contains more detailed guidance and recommendations.
The opinions expressed in this article are those of the authors. They do not purport to reflect the opinions or views of the UKCPA or its members. We encourage readers to follow links and references to primary research papers and guidance.
Competing interest statement:
The author declares: no support from any organisation for the submitted work; RS has been paid for developing and delivering educational presentations from Teva (UK) Ltd; RS has received sponsorship from Teva (UK) Ltd and Napp Pharmaceuticals Ltd to attend educational meetings; RS has attended educational meetings sponsored by Boehringer Ingelheim and Roche; RS has participated in advisory meetings organised by Napp Pharmaceuticals Ltd and Teva (UK) Ltd; RS is a member of the NICE Chronic Obstructive Pulmonary Disease (COPD) Guideline Committee.




