Antimicrobial use in maternity units often occurs outside of the watchful eye of the Antimicrobial Stewardship (AMS) teams. Lower rates of antimicrobial resistance colonisation, lower C.difficile infection rates and the homogeneity of infection presentations see a relative uniformity of prescribing, usually in line with local guidelines, and thus operates outside the purview of stretched AMS teams. Yet maternal sepsis remains one of the leading causes of maternal death in the UK and rates appear to be increasing.
Risk factors for maternal sepsis can be easily recognised, including caesarean section and operative vaginal birth (forceps or vacuum extraction). Without prophylaxis, an estimated 20 to 25 percent of women have an infection following caesarean birth, and up to 16 percent have an infection following operative vaginal birth. Whilst surgical antimicrobial prophylaxis for caesarean birth is well established within the UK, sufficient evidence is not available to support operative vaginal birth antimicrobial prophylaxis.
A welcome addition to the sparse evidence base
A UK-based study recently published in the Lancet aimed to demonstrate the role of antimicrobial prophylaxis in operative vaginal birth in a clinical setting that reflects UK practice.
The multicentre, randomised, blinded, controlled trial was completed at 27 hospital obstetric units in the UK between March 2016 and June 2018. It randomised (1:1) women undergoing operative vaginal delivery (with forceps or vacuum) to receive a single dose of co-amoxiclav 1.2g or placebo (saline solution).
Whilst it certainly will reduce post-partum antimicrobial prescribing, its role in reducing severe sepsis is less clear.
Sensible inclusion and exclusion criteria were used: patients receiving antimicrobials prior to or during delivery were included in the study, patients requiring post-operative antimicrobials for delivery related infections were not. All women with third- and fourth-degree tears were also excluded. Due to the study drug choice, penicillin allergic patients were excluded.
The study was powered to detect a 50 percent reduction in infection complications assuming a four percent prevalence of peri-operative infections, despite the author acknowledging infection rates of near 16 percent in external studies. Acceptable power calculations were included based on this assumption (n=1712 in each arm).
The primary outcome was confirmed or suspected maternal infection within six weeks of delivery. The definition of this outcome is of importance. This includes: a) microbiologically confirmed postpartum infection (objective); b) endometritis as defined by US CDCC criteria (objective); and c) any antimicrobial usage for suspected post-partum infection (somewhat subjective).
Secondary outcomes such as SIRS defined sepsis, PHE defined post-op wound infections and a qualitative questionnaire on patient-report health status was also included. Enrolled patients were followed up meticulously and thus a low loss of participants was evident (less than three percent of the total study population). However, primary outcomes were missing in seven percent of patients.
Results show improvements in the intervention group
The results of the trial show a clear reduction in the proportion of women who had a confirmed or suspected infection after operative vaginal birth in the intervention group, as well as lower proportions of women with perineal wound infection, perineal pain, or perineal wound breakdown after antibiotic prophylaxis.
The rates of confirmed or suspected infection (11 percent in the treatment arm versus 19 percent in the placebo arm [RR 0.58, 95% CI 0.49 – 0.69, p<0.0001]) are higher than expected in the study design (bearing in mind that four percent was used in the power calculation), but ultimately this demonstrates a significant improvement in the primary outcome. On closer investigation, objective measures of post-partum sepsis such as confirmed systemic infection on culture and CDCC diagnosed endometritis are rarely diagnosed within the study (less than one percent). Thus, the number needed to treat (NNT) for reducing objective measures of severe infection (culture positive sepsis, endometritis, SIRS systemic sepsis and deep incisional infections) are high (see table 1 below).
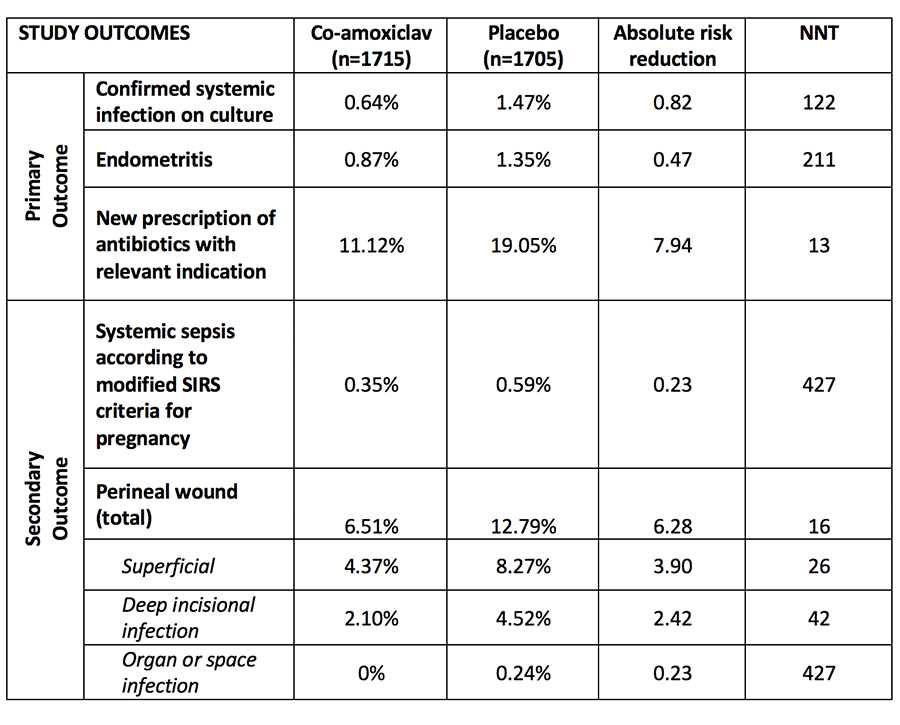
Most of the ‘benefits’ seen with this intervention was with a reduction in antimicrobial prescribing for suspected infection. The indication for these antimicrobials is unclear as they do not appear to correlate to the rates of culture positive systemic infections, CDCC diagnosed endometritis or SIRS diagnoses systemic sepsis. Perineal related infection rates of almost 13 percent in the placebo group (vs 6.51%, ARR 6.28 NNT 16) likely account for the majority of antimicrobial usage in this study. This may reflect the high rates of episiotomy rates seen in this study (89 percent) with approximately another eight percent of women having a perineal tear during operative delivery (remember third- and fourth-degree tears were excluded).
The published article does not allow readers to identify outcomes of episiotomy intervention related to antimicrobial usage but due to the almost ubiquitous use of episiotomy, we can make the tangible links to this practice and increased perineal wounds post-partum.
Secondary outcomes, such as patient reported perineal pain, wound breakdown, primary care or home visits by a midwife and out-patient visits, are all statistically improved in the intervention group with much more appealing NNT expected. Economic savings of reduced healthcare follow up within the intervention group is reported as an average of £52 per patient (99% CI £115.10 to -£9.90).
What are the implications for antimicrobial resistance?
The study demonstrates that, on average, total antimicrobial use is lower in the intervention group with a NNT of 13 women with prophylaxis to reduce one treatment course of antimicrobials post-partum. The impact of population-level antimicrobial resistance through the degree of selective pressure of a single dose co-amoxiclav to all versus a prolonged treatment course to an additional 7.94% of the population is unclear. Short course or single dose prophylaxis should be promoted as we expect a reduced selective pressure on the microbiome. But the number needed to harm (NNH) is unknown; is it greater or less than the NNT presented in this study? Does a stitch in time save nine?
Also of interest to AMS practitioners is both the choice of antimicrobial and the timing of administration. The study authors were open in the analysis of drug choice, with a second generation cephalosporin with metronidazole not chosen as it remains off formulary in many centres due to perceived C.difficile complication risk. Co-amoxiclav offers similar coverage with the advantage of enterococcal cover (a possible pathogen for perineal infection) and more robust Group A strep coverage (a major pathogen associated with maternal death). What we offer to our penicillin intolerant or allergic women is less clear and will be discussed within AMS teams across the country. High rates of macrolide / lincosamide resistance makes clindamycin less attractive despite its appeal for preventing perineal infections, and quinolones or tetracyclines are unlikely to be popular or suitable in breastfeeding women.
The timing of dosing postpartum (median 3.2 hours) is novel but pragmatic. Pre-intervention dosing for this unpredictable operative procedure is problematic. Whether appropriately dosed oral treatment is sufficient will be discussed by AMS and obstetric teams across the country; whilst this is an attractive option, we simply do not currently have the supporting evidence, but we expect many to explore this option in the future.
Conclusion
This well-designed UK study is a welcome addition to the sparse evidence base on antimicrobial use in operative vaginal births. The headline news will be that a single dose co-amoxiclav reduces operative vaginal birth infective complications. Whilst it certainly will reduce post-partum antimicrobial prescribing and superficial and deep incisional infections, its role in reducing severe sepsis and thus preventing maternal death is less clear.
It poses some interesting questions for AMS teams to ponder, including the benefits of single dose antimicrobials for all as a means to reduce total antimicrobial exposure, the options for penicillin intolerant patients, and how we balance AMS goals versus healthcare economic benefits.
The published study can be found here:
Knight M. et al. Prophylactic antibiotics in the prevention of infection after operative vaginal delivery (ANODE): a multicentre randomised controlled trial. The Lancet 2019; 393: 2395–403.
The opinions expressed in this article are those of the authors. They do not purport to reflect the opinions or views of the UKCPA or its members. We encourage readers to follow links and references to primary research papers and guidance.
Competing interest statement:
The author declares: no support from any organisation for the submitted work; no financial relationships with any organisations that might have an interest in the submitted work in the previous three years; no other relationships or activities that could appear to have influenced the submitted work.




